HA Pharmacology & Pharmacy – Unit 4
Drugs Acting on Central Nervous System – CTEVT Second Year Notes
According to CTEVT Syllabus (2024) | Health Science (PCL)
Introduction to CNS Drugs
Drugs acting on the Central Nervous System (CNS) are among the most clinically important medications, used to manage conditions ranging from anxiety and epilepsy to surgical anesthesia and psychiatric disorders. This unit covers the pharmacology of drugs that alter brain function, including their mechanisms, therapeutic uses, and adverse effects.
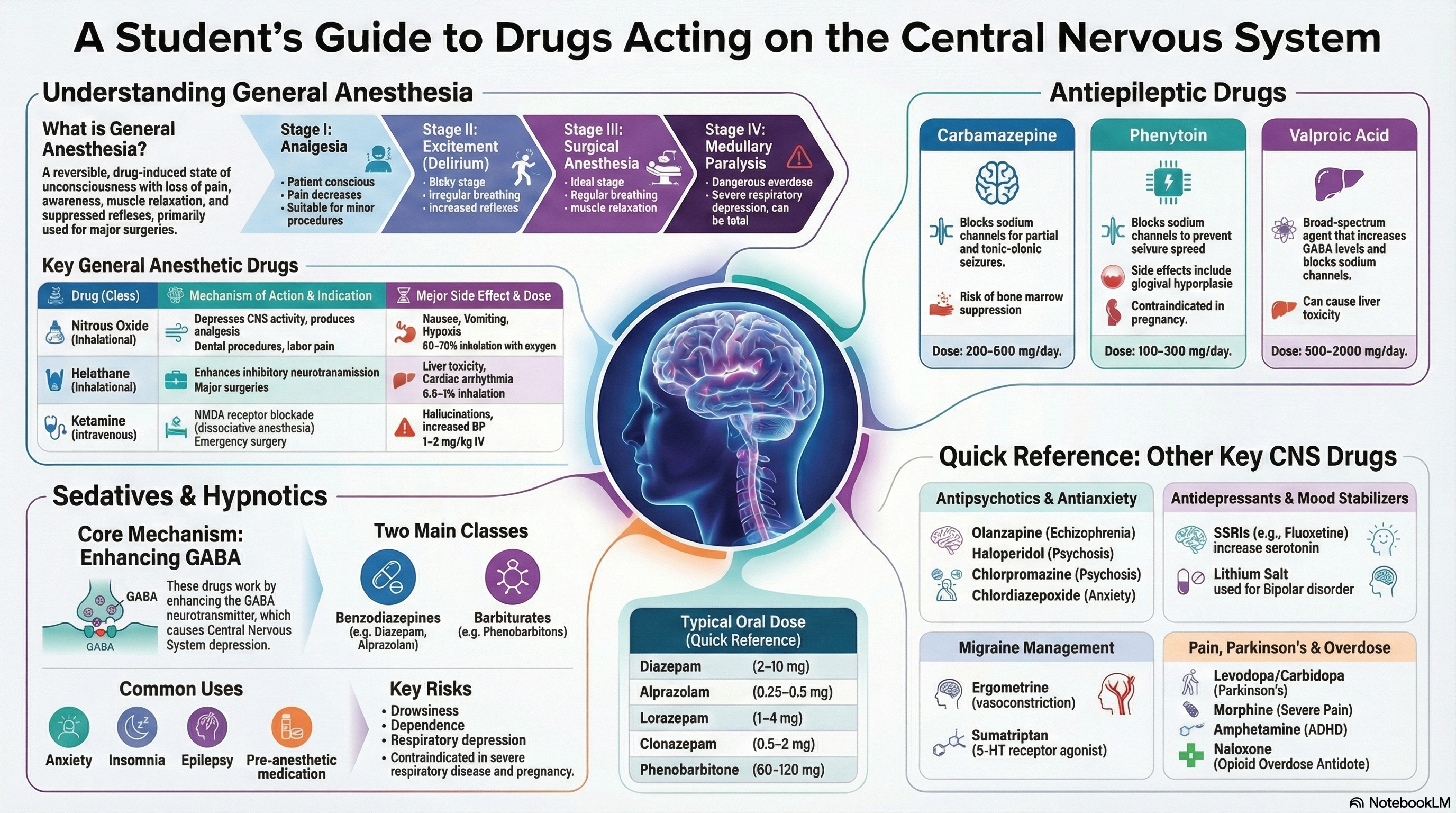
1. General Anesthesia
Definition & Goals
General anesthesia is a reversible, drug-induced state characterized by:
Loss of awareness
Absence of pain perception
No memory of procedure
For surgical access
Autonomic stability
Stages of General Anesthesia (Guedel’s Classification)
| Stage | Name | Characteristics | Clinical Significance | Duration |
|---|---|---|---|---|
| Stage I | Analgesia | Conscious, pain perception reduced, hearing exaggerated, amnesia begins | Minor procedures (dental, wound suturing) | From induction to loss of consciousness |
| Stage II | Excitement/Delirium | Unconscious but reflexive, irregular breathing, vomiting, pupil dilation, increased muscle tone | Dangerous stage – avoid stimulation, risk of aspiration | Brief, should be minimized |
| Stage III | Surgical Anesthesia | Regular breathing, pupil constriction, progressive muscle relaxation, absent reflexes | Ideal for surgery, divided into 4 planes of increasing depth | Maintained throughout surgery |
| Stage IV | Medullary Paralysis | Respiratory arrest,瞳孔 dilation, cardiovascular collapse | Overdose – requires immediate resuscitation | Fatal if prolonged |
General Anesthetic Drugs
Nitrous Oxide (N₂O)
Class: Inhalational anesthetic
Mechanism: NMDA receptor antagonism, weak anesthetic but potent analgesic
Indications: Dental procedures, labor analgesia, minor surgery (adjunct)
Dose: 30-70% with oxygen (never 100%)
Halothane
Class: Inhalational anesthetic (halogenated hydrocarbon)
Mechanism: Enhances GABAₐ and glycine receptors, inhibits NMDA
Indications: Induction (especially pediatric) and maintenance of anesthesia
Dose: Induction 2-4%, Maintenance 0.5-1.5%
Ketamine
Class: IV anesthetic (arylcyclohexylamine)
Mechanism: Noncompetitive NMDA receptor antagonist → dissociative anesthesia
Indications: Emergency/trauma surgery, pediatric procedures, burns dressing, refractory depression
Dose: IV: 1-2 mg/kg, IM: 4-6 mg/kg (premedicate with benzodiazepine)
📌 Clinical Tips: General Anesthesia
- Balanced anesthesia: Combination of drugs to achieve all components (e.g., opioid + muscle relaxant + inhalational agent)
- Minimum Alveolar Concentration (MAC): Measure of potency – lower MAC = more potent
- Malignant hyperthermia: Life-threatening reaction to volatile anesthetics – treat with dantrolene
- Pre-anesthetic medication: Benzodiazepines (anxiety), anticholinergics (reduce secretions), antiemetics
2. Sedatives-Hypnotics
Benzodiazepines
Mechanism: Potentiate GABAₐ receptor → ↑ chloride influx → neuronal hyperpolarization
| Drug | Half-life | Key Uses | Adult Oral Dose | Special Notes |
|---|---|---|---|---|
| Diazepam | 20-50 hours | Anxiety, muscle spasm, alcohol withdrawal, status epilepticus (IV) | 2-10 mg 1-4× daily | Active metabolites, IV formulation irritant |
| Alprazolam | 6-12 hours | Panic disorder, generalized anxiety | 0.25-0.5 mg 2-3× daily | High abuse potential, difficult withdrawal |
| Lorazepam | 10-20 hours | Anxiety, pre-anesthetic, status epilepticus | 1-4 mg/day in divided doses | No active metabolites, preferred in liver disease |
| Clonazepam | 18-50 hours | Epilepsy (absence, myoclonic), panic disorder | 0.5-2 mg/day (max 20 mg) | Long-acting, tolerance develops to anticonvulsant effect |
Barbiturates (Phenobarbitone)
Mechanism: Prolong GABAₐ receptor opening → stronger effect than benzodiazepines
Uses: Epilepsy (generalized tonic-clonic, partial), neonatal seizures, status epilepticus
Dose: 60-120 mg/day oral (therapeutic level: 15-40 μg/mL)
3. Antiepileptic Drugs (AEDs)
| Drug | Mechanism | Primary Seizure Types | Key Side Effects | Special Considerations | Dose Range |
|---|---|---|---|---|---|
| Carbamazepine | Blocks voltage-gated Na⁺ channels, ↓ high-frequency firing | • Partial (focal) seizures • Generalized tonic-clonic • Trigeminal neuralgia |
• Dizziness, diplopia • Hyponatremia (SIADH) • Rash (SJS risk) • Bone marrow suppression |
• Autoinduction (dose ↑ over weeks) • Teratogenic (neural tube defects) • Check Na⁺, CBC, LFT |
200-600 mg BD Therapeutic: 4-12 μg/mL |
| Phenytoin | Use-dependent Na⁺ channel blockade, inhibits glutamate release | • Generalized tonic-clonic • Partial seizures • Status epilepticus (IV) |
• Gingival hyperplasia • Hirsutism, coarsening • Ataxia, nystagmus • Megaloblastic anemia |
• Non-linear kinetics (small dose ↑ → large level ↑) • IV: cardiac monitoring (risk of arrhythmia) • Fetal hydantoin syndrome |
100-300 mg/day Therapeutic: 10-20 μg/mL |
| Valproic Acid (Sodium Valproate) | • ↑ GABA synthesis/inhibition of breakdown • Blocks Na⁺ channels • T-type Ca²⁺ channel blockade |
• Broad spectrum • Absence seizures • Myoclonic, tonic-clonic • Bipolar disorder, migraine prophylaxis |
• Weight gain, tremor • Hair loss (transient) • Hepatotoxicity (children <2 yr) • Pancreatitis, thrombocytopenia |
• High teratogenic risk (neural tube defects) • Check LFT, platelets • Enteric-coated to reduce GI upset |
500-2000 mg/day Therapeutic: 50-100 μg/mL |
📌 Clinical Guidelines for AEDs
4. Antipsychotics & Antidepressants
Antipsychotics
Typical (First-Generation)
Mechanism: D₂ receptor antagonism in mesolimbic pathway
Examples: Haloperidol, Chlorpromazine, Fluphenazine
Atypical (Second-Generation)
Mechanism: 5-HT₂ₐ > D₂ antagonism, some with partial D₂ agonism (aripiprazole)
Examples: Olanzapine, Risperidone, Quetiapine, Clozapine
Key Drugs
Olanzapine: 5-20 mg/day, for schizophrenia, bipolar. High metabolic risk.
Haloperidol: 1-10 mg/day, acute psychosis, IV for delirium. High EPS risk.
Chlorpromazine: 100-400 mg/day, schizophrenia, antiemetic. Sedating.
Antidepressants
| Class | Mechanism | Examples & Doses | Key Side Effects | Clinical Notes |
|---|---|---|---|---|
| SSRIs (Selective Serotonin Reuptake Inhibitors) |
Block SERT → ↑ synaptic serotonin | • Fluoxetine (20-40 mg/day) • Escitalopram (10-20 mg/day) • Paroxetine (20-40 mg/day) • Sertraline (50-150 mg/day) |
• Nausea, headache • Sexual dysfunction • Serotonin syndrome (risk with MAOIs) • Withdrawal on discontinuation |
First-line for depression, anxiety. Delayed onset (2-4 weeks). Fluoxetine long half-life (7-15 days). |
| TCAs (Tricyclic Antidepressants) |
Block NET & SERT → ↑ NE & 5-HT, also anticholinergic, antihistaminic, α-blocking | • Amitriptyline (25-150 mg/day) • Imipramine • Clomipramine |
• Anticholinergic (dry mouth, constipation, urinary retention) • Sedation, weight gain • Cardiac arrhythmias (overdose fatal) • Orthostatic hypotension |
Used in neuropathic pain, migraine prophylaxis. Lethal in overdose. Therapeutic window. |
| Other | Various mechanisms | • Venlafaxine (SNRI) • Bupropion (NE/DA reuptake) • Mirtazapine (α₂ antagonist) |
Class-dependent: hypertension (venlafaxine), seizures (bupropion), weight gain (mirtazapine) | Second-line or when specific side effect profile needed (e.g., bupropion for sexual dysfunction). |
Mood Stabilizer: Lithium
Indications: Bipolar disorder (acute mania & maintenance), augmentation in depression
Mechanism: Inhibits inositol monophosphatase, modulates glutamate/GABA
Dose: 600-1200 mg/day, target level 0.6-1.2 mEq/L (acute: 0.8-1.2, maintenance: 0.6-0.8)
5. Migraine Management & Other CNS Drugs
Migraine-Specific Drugs
Triptans (Sumatriptan)
Mechanism: 5-HT1B/1D agonist → cranial vasoconstriction, inhibits neurogenic inflammation
Use: Acute migraine attack (not for prophylaxis)
Dose: 50-100 mg oral, 6 mg SC, 20 mg nasal spray
Ergot Alkaloids (Ergometrine)
Mechanism: Nonselective 5-HT agonist, also α-adrenergic/dopaminergic effects
Uses: Migraine (less preferred), postpartum hemorrhage (uterine contraction)
Dose: 0.5-1 mg IM/IV for PPH; migraine: ergotamine 1-2 mg oral
| Drug Class | Drug | Primary Use | Key Mechanism | Dose | Important Notes |
|---|---|---|---|---|---|
| Parkinson’s | Levodopa + Carbidopa | Parkinson’s disease | Dopamine precursor + peripheral DDC inhibitor | Levodopa: 250-1000 mg/day Carbidopa: 25-100 mg/day |
“On-off” phenomena, dyskinesias, never give with MAOIs |
| Opioid Analgesics | Morphine | Severe pain (cancer, MI, trauma) | μ-opioid receptor agonist | 5-10 mg IM/SC q4h 2.5-5 mg IV |
Respiratory depression, constipation, miosis, addiction potential |
| Opioid Analgesics | Pethidine (Meperidine) | Moderate-severe pain, labor | μ-opioid agonist, anticholinergic | 50-100 mg IM q3-4h | Active metabolite normeperidine → seizures with renal impairment |
| Opioid Antagonist | Naloxone | Opioid overdose reversal | Competitive opioid receptor antagonist | 0.4-2 mg IV/IM/SC, repeat q2-3min | Short half-life (1-2h) → monitor for renarcotization, can precipitate withdrawal |
| Stimulant | Amphetamine | ADHD, narcolepsy | ↑ NE/DA release, reuptake inhibition | 5-30 mg/day in divided doses | High abuse potential, insomnia, anorexia, cardiovascular effects |
One Page Summary: CNS Drugs
• Stages: I (Analgesia), II (Excitement), III (Surgical), IV (Medullary paralysis)
• Nitrous oxide: Dental, labor pain, diffusion hypoxia
• Halothane: Hepatotoxicity, arrhythmias
• Ketamine: Dissociative anesthesia, hallucinations, ↑ BPSEDATIVES-HYPNOTICS
• Benzodiazepines: GABA potentiation, anxiolytic, hypnotic, muscle relaxant
• Diazepam: Long-acting, active metabolites
• Lorazepam: No active metabolites, status epilepticus
• Phenobarbitone: Enzyme inducer, respiratory depression risk
ANTIEPILEPTICS
• Carbamazepine: Na⁺ channel blocker, hyponatremia, SJS risk
• Phenytoin: Gingival hyperplasia, non-linear kinetics
• Valproate: Broad spectrum, hepatotoxicity, teratogenic
ANTIPSYCHOTICS
• Typical: D₂ antagonists → EPS (haloperidol)
• Atypical: 5-HT₂ₐ > D₂ → metabolic effects (olanzapine)
• Olanzapine: 5-20 mg/day, weight gain
• Haloperidol: 1-10 mg/day, high EPS
• SSRIs: First-line (fluoxetine, escitalopram), sexual dysfunction
• TCAs: Amitriptyline, anticholinergic side effects, lethal in overdose
• Lithium: Bipolar, narrow therapeutic index (0.6-1.2 mEq/L), monitor levelsMIGRAINE
• Triptans (sumatriptan): 5-HT1B/1D agonists, acute attack
• Ergots: Vasoconstrictors, ergotism risk
OTHER CNS DRUGS
• Levodopa/carbidopa: Parkinson’s, on-off phenomena
• Morphine: μ-opioid agonist, respiratory depression
• Pethidine: Metabolite causes seizures
• Naloxone: Opioid overdose antidote, short half-life
• Amphetamine: ADHD, stimulant, abuse potential
MNEMONICS
• Benzodiazepine reversal: Flumazenil
• Phenytoin side effects: Hirsutism, Hyperplasia, Hematologic, Ataxia
• SSRIs side effects: Sexual dysfunction, Sleep disturbance, Serotonin syndrome
Monitor drug levels for phenytoin, lithium, valproate. Know reversal agents: flumazenil (BZDs), naloxone (opioids).
Clinical Application Scenarios
Scenario 1: Status Epilepticus
Presentation: 25-year-old with continuous tonic-clonic seizures for 10 minutes.
Immediate Management: ABCs, IV access, glucose check.
Drug Sequence: Lorazepam 4 mg IV → If persists: Phenytoin/fosphenytoin loading → Refractory: midazolam infusion, propofol.
Monitoring: Airway, respiratory status, ECG with phenytoin.
Scenario 2: Opioid Overdose
Presentation: Unconscious, pinpoint pupils, respiratory rate 6/min, SpO₂ 85%.
Diagnosis: Opioid toxicity (heroin/morphine overdose).
Treatment: Naloxone 0.4-2 mg IV/IM, repeat q2-3min until breathing adequate.
Key: Monitor for renarcotization (naloxone half-life shorter than opioids).
Scenario 3: Acute Migraine
Presentation: 30-year-old with unilateral throbbing headache, nausea, photophobia.
First-line: NSAIDs (ibuprofen) + antiemetic (metoclopramide).
If severe: Triptan (sumatriptan 50-100 mg oral).
Contraindications: Avoid triptans if history of stroke, CAD, hemiplegic migraine.
Topic Tags
Benzodiazepines
Antiepileptic Drugs
Antipsychotics
Antidepressants
SSRIs
Lithium
Migraine Treatment
Opioids
Parkinson’s Drugs
Ketamine
Phenytoin
Valproic Acid
Naloxone
Sumatriptan
CTEVT Syllabus
Health Assistant
CNS Pharmacology
Psychiatric Drugs
HA Second Year
Key Clinical Takeaways
- General anesthesia stages: Aim for Stage III, avoid Stage II (excitement) and IV (overdose).
- Benzodiazepines enhance GABA; flumazenil reverses overdose. Avoid abrupt discontinuation.
- Antiepileptic drug choice depends on seizure type: carbamazepine for partial, valproate for generalized.
- Antipsychotics: Typical cause EPS, atypical cause metabolic syndrome. Clozapine for treatment-resistant but requires CBC monitoring.
- SSRIs are first-line antidepressants but have delayed onset (2-4 weeks) and sexual side effects.
- Opioid overdose: triad of coma, pinpoint pupils, respiratory depression; treat with naloxone.
Quick Self-Check
Question 1: Which stage of general anesthesia is characterized by irregular breathing and increased reflexes?
Question 2: Match the drug to its reversal agent:
1. Benzodiazepines
2. Opioids
3. Heparin
Download Notes
Get a printable PDF version of these comprehensive CNS pharmacology notes. HA Pharmacology and Pharmacy Unit 4 Drugs Acting on Central Nervous System Notes
Includes all drug classes, mechanisms, doses, and clinical scenarios. HA Pharmacology and Pharmacy Unit 4 Drugs Acting on Central Nervous System Notes
Further Reading & Resources
- Official CTEVT Syllabus: CTEVT Website
- Epilepsy Foundation: Epilepsy Treatment Guidelines
- American Psychiatric Association: APA Practice Guidelines


