Differentiate Between Saturated and Unsaturated Vapour
Differentiate Between Saturated and Unsaturated Vapour – In physics, vapour can exist in different states depending upon temperature and humidity inside a container. The comparison helps us understand evaporation and condensation.
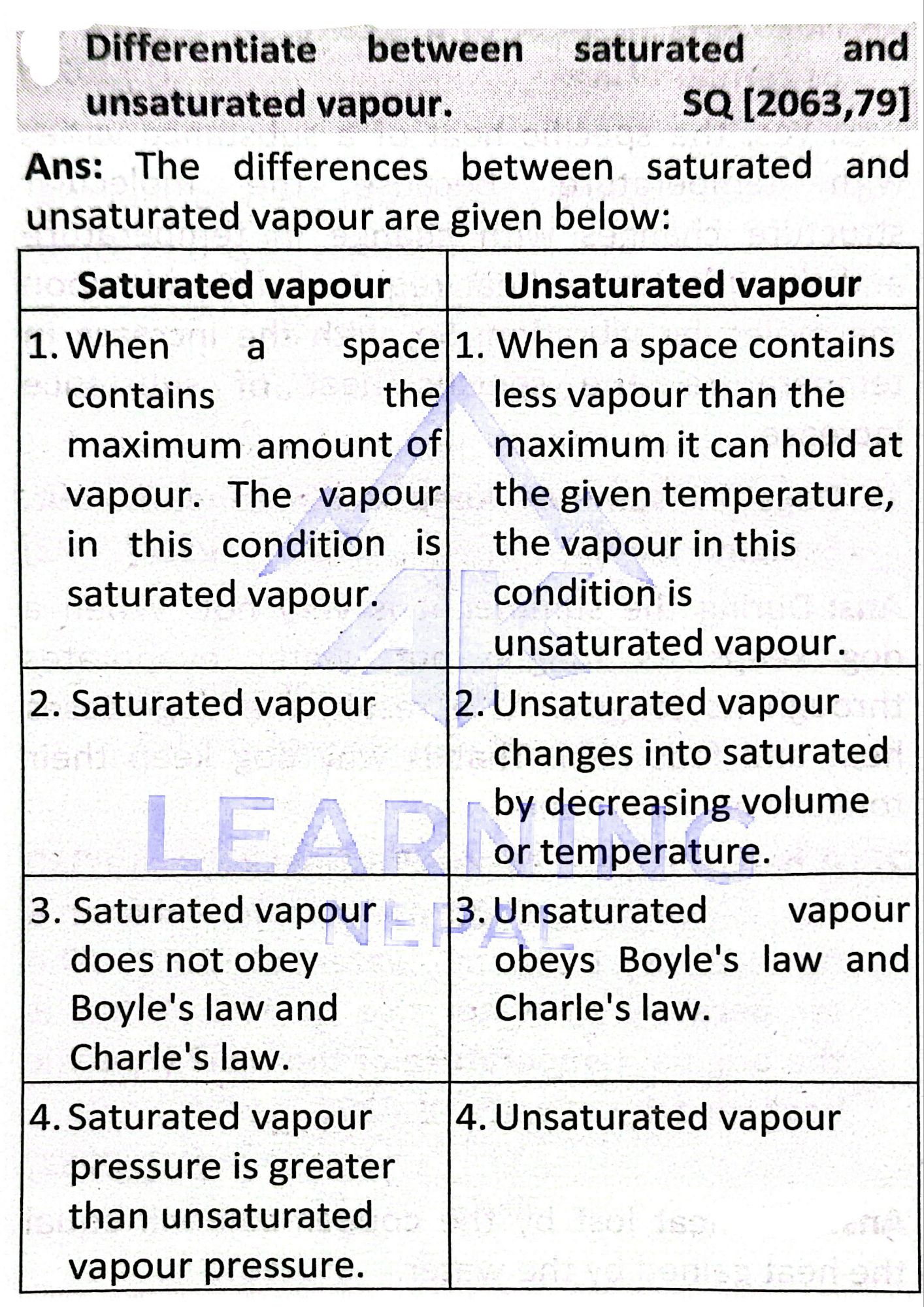
Saturated Vapour
When vapour contains maximum possible amount of vapour at particular temperature, it is called saturated vapour.
Unsaturated Vapour
When vapour still can take more vapour molecules and is not fully filled, it is known as unsaturated vapour.
Difference Table
| Saturated Vapour | Unsaturated Vapour |
|---|---|
| Contains maximum vapour | Still contains less than maximum |
| No further evaporation | More evaporation possible |
| Occurs at high humidity | Occurs at low humidity |
Note: Evaporation continues until vapour becomes saturated inside a closed vessel.
Related: Internal link here
Differentiate Between Saturated and Unsaturated Vapour
Keywords
saturated vapour, unsaturated vapour, physics vapour, evaporation, condensation, vapour pressure, temperature vapour, humidity, closed vessel vapour, vapour formation, class 10 physics, saturated definition, unsaturated meaning, difference vapour, physics concept,Differentiate Between Saturated and Unsaturated Vapour
Tags
saturated vapour, unsaturated vapour, class 10 physics, vapour difference, chapter heat, evaporation topic, physics notes, cbse science, temperature physics, vapour comparison, nepali class 10, science notes, physics question, vapour table, exam question,Differentiate Between Saturated and Unsaturated Vapour

